Two studies from the Immunology group on autoimmunity and hematopoietic stem cells show promising therapeutic potential

Ahead Therapeutics team
Searching for a strategy to safely and definitively restore self-tolerance in autoimmune diseases
The study in mice published in the journal Nanomedicine: Nanotechnology, Biology and Medicine proposes a therapy based on the therapeutic efficacy of phosphatidylserine (PS) liposomes. Previously, they have been used to encapsulate self-antigens and treat experimental type 1 diabetes and multiple sclerosis and, in this research, they are adapted to other autoimmune diseases (AIDs) such as rheumatoid arthritis and myasthenia gravis with the replacement of the encapsulated self-antigen.
The work reinforces the capacity of autoantigen-loaded PS-liposomes as a preventive and therapeutic platform for AIDs, showing advantages for future clinical use. The joint action of PS and autoantigen results in a tolerogenic effect on immune cells, which activate the machinery capable of arresting autoimmunity in a specific manner.
Autoimmune diseases are a major health problem in developed countries. There are more than 80 of them and their incidence is increasing. Alternatives to standard treatments, which often have side effects, are being sought. However, the antigen-specific immunotherapies tested so far, aimed at stopping the autoimmune attack, have had disappointing results. The present study addresses the problem and shows a specific, safe and long-lasting platform that could be used in various autoimmune diseases.
This work is the result of a collaboration between several centres. Led by the spin-off Ahead Therapeutics, it involved the Immunology of Diabetes group of the Germans Trias i Pujol Research Institute (IGTP), and researchers from the Biomedical Research Institute of Lleida (IRBLleida), the Friedrich-Alexander-Universität Erlangen-Nürnberg (Germany), the Institut de Myologie (INSERM, France) and Orgentec Diagnostika (Germany).
The principal investigator of the study, Dr Marta Vives-Pi, leader of the IGTP Immunology of Diabetes research group and scientific director of Ahead Therapeutics, says that "this work describes a platform for tolerance induction and treatment of autoimmune diseases that is specific, biocompatible and with long-term effects. We have determined the mechanism of action of the therapy, which is based on a physiological process and works preventively and therapeutically."
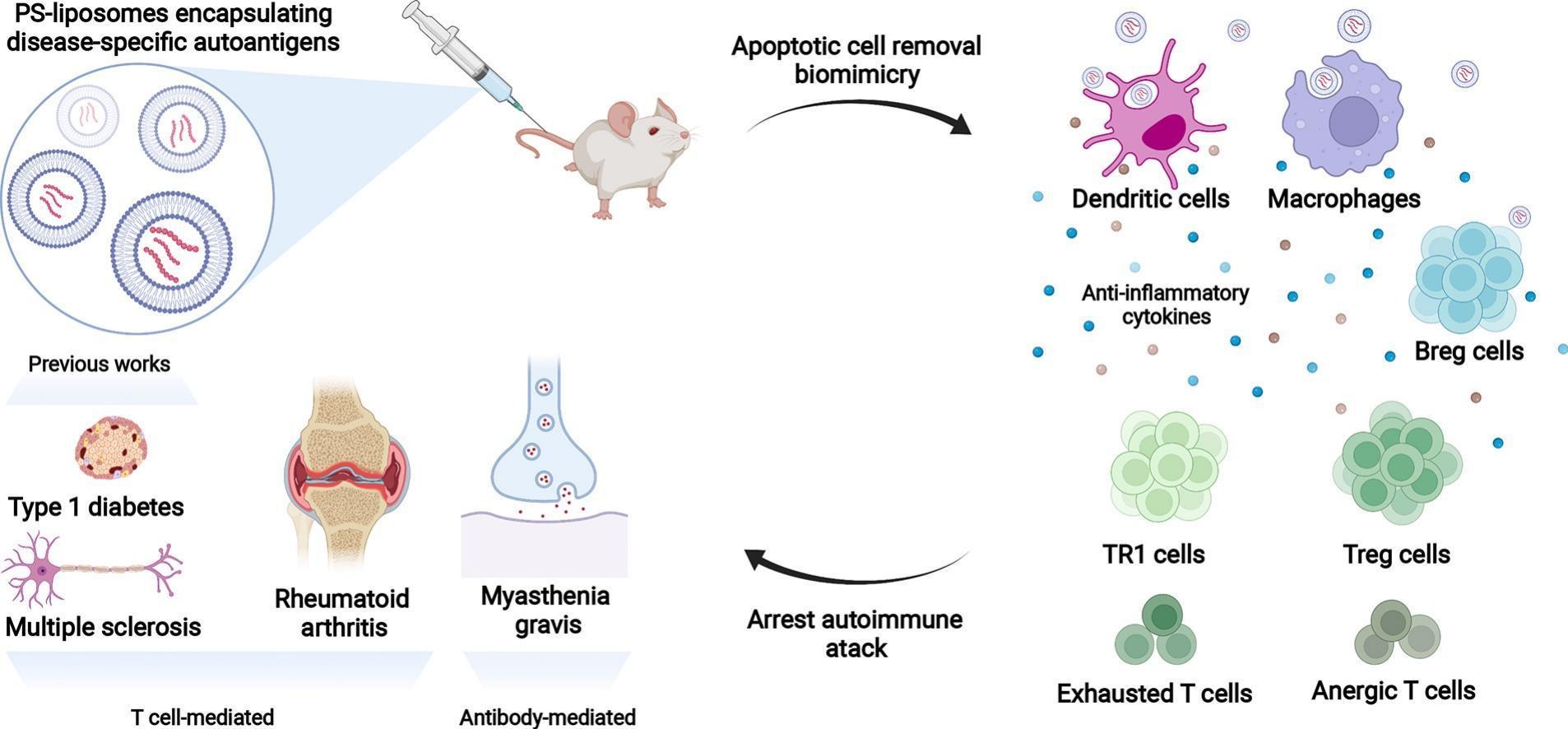
Graphic summary. Created with Biorender.
Making the most of umbilical cord blood cells, key against hematologic diseases
Another recent study from the IGTP Immunology of Diabetes group provides a strategy to improve the engraftment capabilities of umbilical cord blood stem cells. It has been published in Archivum Immunologiae et Therapiae Experimentalis and involved researchers from the IGTP, the University Medical Center Hamburg-Eppendorf (UKE), the Josep Carreras Leukemia Research Institute (IJC) and the Blood and Tissue Bank (BST).
Hematopoietic stem cell (HSC) transplantation is vital for the treatment of hematologic and genetic diseases. The main sources of stem cells are bone marrow and mobilized peripheral blood. Another possibility is umbilical cord blood, with less impact on the donor. The limitation in this case is that the number of stem cells is low, which causes 90% of umbilical cords to be discarded. This work seeks a strategy to optimize transplantation with few stem cells, by pre-treatment with a synthetic glucocorticoid, betamethasone.
This is the first study to demonstrate that betamethasone modifies the phenotype of cord blood CD34+ HSCs and improves their engraftment capacity in an experimental model. This leads to faster recovery of the immune system and promotes the subset of Treg lymphocytes (which regulate or suppress other immune system cells) that contribute to the survival of engrafted cells. Further research will be needed for the finding to have a clinical impact, but the results are encouraging and show the benefits of betamethasone in HSC transplantation.
The first author of the article, David Perna-Barrull, explains that "pre-treatment of HSCs with betamethasone would make it possible to use umbilical cord units, which until now have been discarded. This would mean having more compatible donors available for hematopoietic stem cell transplantation".
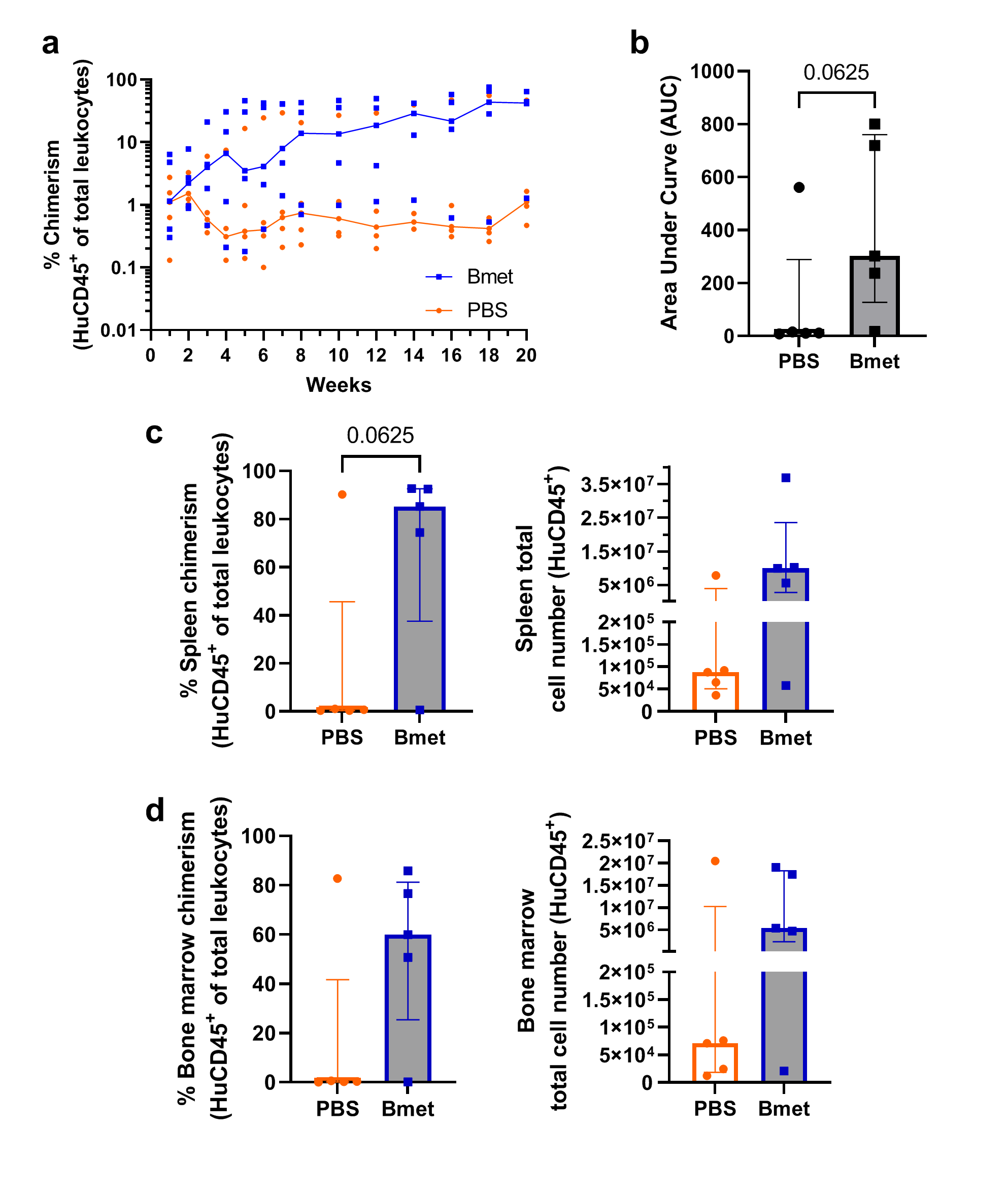
Figure 4a of the study. Percentage of human immune system cells expanded in vivo (mouse) after pre-treatment with betamethasone and during 20 weeks post-transplantation (in blue) compared to those not exposed to treatment (in orange).
References
Almenara-Fuentes, L, Rodriguez-Fernandez, S, Rosell-Mases, E., Kachler, K, You, A, Salvado, M, Andreev, D, Steffen, U, Bang, H, Bozec, A, Schett, G, Le Panse, R, Verdaguer, J, Dalmases, M, Rodriguez-Vidal, S, Barneda-Zahonero, B, & Vives-Pi, M. A new platform for autoimmune diseases. Inducing tolerance with liposomes encapsulating autoantigens. Nanomedicine: nanotechnology, biology, and medicine. 2022 Dec 5;48:102635. DOI: https://doi.org/10.1016/j.nano.2022.102635
Perna-Barrull, D, Gomez-Muñoz, L, Rodriguez-Fernandez, S, Gieras, A, Ampudia-Carrasco, R M, Almenara-Fuentes, L, Risueño, R M, Querol, S, Tolosa, E, & Vives-Pi, M. Impact of Betamethasone Pretreatment on Engrafment of Cord Blood-Derived Hematopoietic Stem Cells. Archivum immunologiae et therapiae experimentalis. 2022 Dec 18;71(1):1. DOI: https://doi.org/10.1007/s00005-022-00666-5
