Comprehensive genomic characterisation of malignant peripheral nerve tumour-derived lines challenges current diagnostic criteria
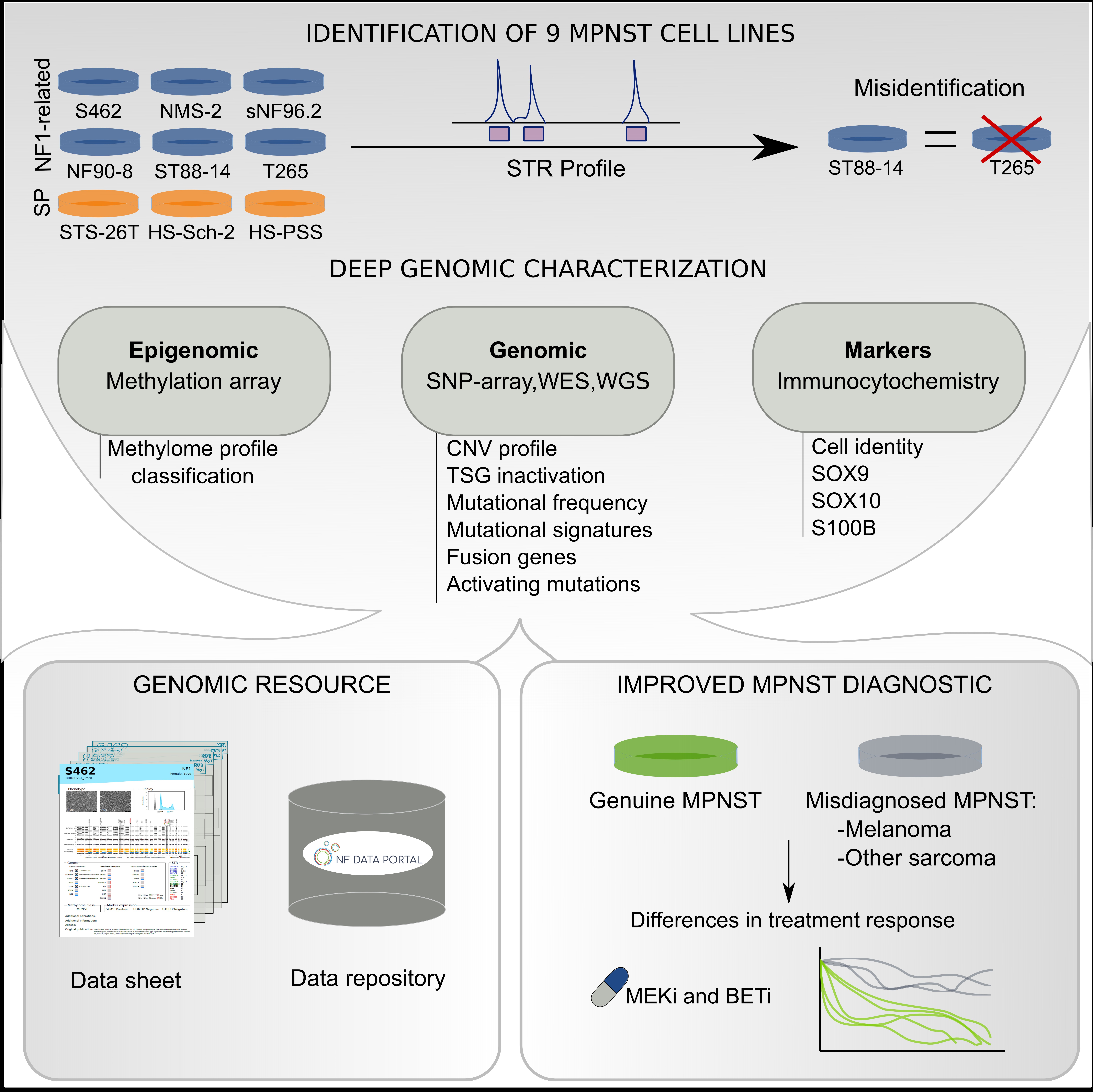
The Hereditary Cancer research group from the Germans Trias i Pujol Research Institute (IGTP) has led an international collaboration that has allowed the genomic characterisation of the most commonly used cell lines derived from malignant peripheral nerve sheath tumours (MPNSTs). This research has generated a detailed catalogue of genomic alterations for each cell line, which can be used to develop strategies for precision therapies. At the same time, a genomic repository has been created that is open to all researchers interested in these tumours. The work has been published in the journal iScience.
One of the most notable results is that it provides new types of genomic information that complements the histological characterisation of MPNSTS for a better and more accurate differential diagnosis of these tumours. In fact, the work has made it possible to identify that some of the lines supposedly MPNSTs, which have been used in multiple research studies, or which are deposited in cell line banks (ATCC, RIKEN), are not MPNSTs, but other tumour entities.
Dr Miriam Magallón, first author of the study, comments "we have identified cell lines that were misdiagnosed, but we have also detected that two of the most commonly used lines as MPNSTs are actually the same cell line".
Drs Bernat Gel and Eduard Serra, the researchers who have led this work, emphasise that "this work is a great opportunity to use the new types of genomic information to revise the diagnostic criteria for these tumour types. We would like to lead a new international project in which pathologists, molecular biologists and bioinformaticians could systematically analyse and compare the histology of these tumours, together with their genomic data, to clarify what an MPNST is, if there are different subtypes, and what is not an MPNST".
Bernat Gel finally highlights "this study also opens the door to the development of new genomic analyses that facilitate the management of patients who are at high risk of developing MPNST, or who have already developed it, and require regular follow-up. Minimal biopsy or liquid biopsy-based analyses using new long-read sequencing technologies would be ideal for identifying the type of genomic alterations responsible for the development and progression of MPNSTs".
The IGTP Hereditary Cancer Group has been studying peripheral nervous system tumours associated with Neurofibromatosis type 1 for years, especially the progression from plexiform neurofibroma (PNF), to atypical neurofibroma (or ANNUBP), to malignant peripheral nerve sheath tumour (MPNST). The two pillars of the characterisation of these tumours by this group are genomic, epigenomic and bioinformatic analyses, on the one hand, and the development of cellular models that reliably represent these tumours, on the other. In relation to this point, the research group has just published a paper in the journal STAR Protocols explaining an experimental protocol for the generation of neurofibrospheres, a 3D model derived from induced pluripotency stem cells (Mazuelas et al. 2023. DOI: 10.1016/j.xpro.2023.102198).
The laboratories responsible for establishing these cell lines, researchers from 12 different laboratories and in 5 different countries (Germany, Belgium, Japan, United States and Spain) have collaborated in this study. The work has been funded by the Instituto de Salud Carlos III (PI17/00524, PI20/00228), the Fundación Proyecto Neurofibromatosis and the Fundació La Marató de TV3 (51/C/2019).
$600,000 grant to conduct a pre-clinical trial of a new therapeutic strategy to treat cutaneous neurofibromas
The research group has received a $615,000 grant from the Neurofibromatosis Therapeutic Acceleration programme (NTAP)-Johns Hopkins University School of Medicine to carry out a project to test a new therapeutic strategy for cutaneous neurofibromas in patients with Neurofibromatosis Type 1. The project is led by the Hereditary Cancer group and is being conducted jointly with the group of Dr Piotr Topilko, from the Mondor Institute for Biomedical Research (France).
People with NF1 can develop from tens to thousands of cutaneous neurofibromas on their skin. These benign tumours, which originate in the nerve endings between the epidermis and dermis, are not life-threatening, but instead have a very high impact on their quality of life. These tumours can be disfiguring, discomforting, and can cover a person's body, significantly affecting self-esteem, socialisation, etc.
The Hereditary Cancer group found that the combination of intracellular cAMP-raising agents, together with MEK (Raso/MAPK pathway) inhibitors, has a major impact on the viability of both primary Schwann cells derived from these tumours and a 3D model of neurofibrospheres derived from induced pluripotent stem cells (iPSCs). With these promising results at the in vitro level, a project was presented within the NTAP initiative "Biology and Therapeutic Development (BTD) for Cutaneous Neurofibromas (cNFs) Application" to make the leap to two complementary animal models and collect pre-clinical in vivo data regarding this co-treatment. One of these models has been developed by Dr Piotr Topilko in Paris and the other is a PDX model that results from grafting neurofibromespheres derived from iPSCs into the sciatic nerve of mice, in collaboration with the group of Dr Conxi Lázaro and Dr Juana Fernández, from the Institut d'Investigació Biomèdica de Bellvitge (IDIBELL).
The project will last three years and will be led by Dr Helena Mazuelas, both in Barcelona and Paris, and coordinated by Dr Meritxell Carrió and Dr Eduard Serra.
Reference
Miriam Magallón-Lorenz et al. Deep genomic analysis of malignant peripheral nerve sheath tumor cell lines challenges current malignant peripheral nerve sheath tumor diagnosis. iScience. 2023 Jan 31;26(2):106096. DOI: 10.1016/j.isci.2023.106096. PMID: 36818284; PMCID: PMC9929861.
