New method for detecting nanoplastics in blood
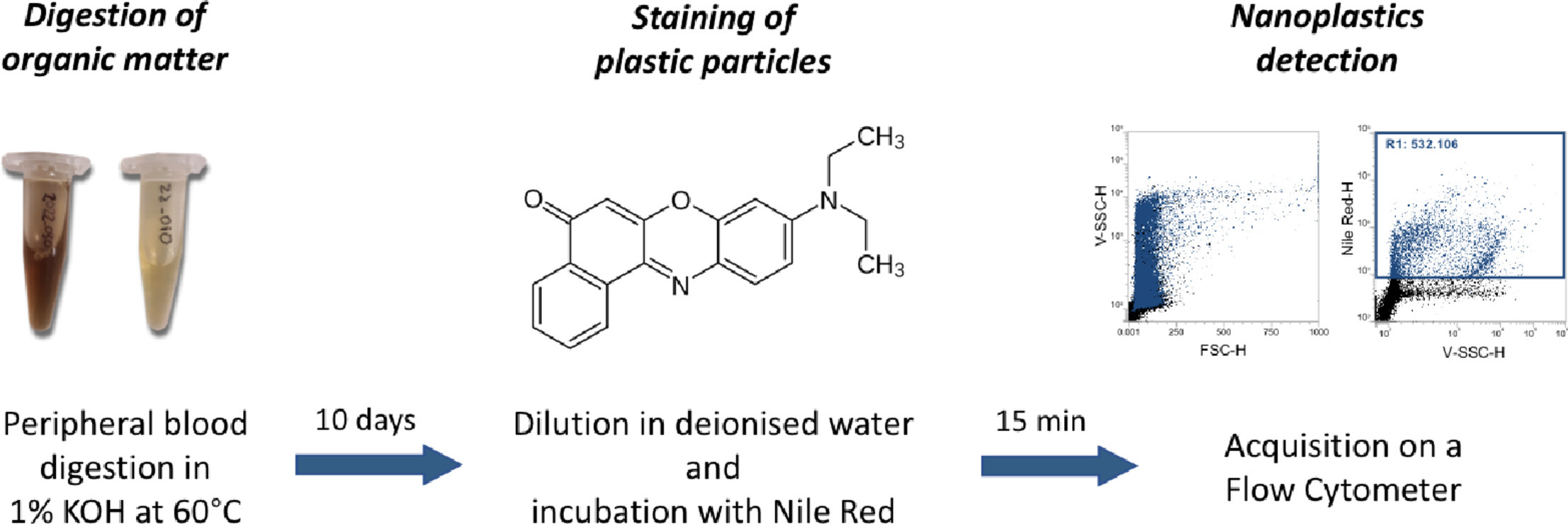
A study uses for the first time flow cytometry to detect and measure nanoplastics in peripheral human blood. People with various medical conditions have been included in the study so as to investigate potential differences for nanoplastic accumulation across the population. In addition, mice have been used in a highly controlled environment to compare their nanoplastic levels to that of humans. This study, which has been published at the journal MethodsX.





_1676552884.png)



